-
Since 1996
Premier Medical
is one of the worldwide leaders in developing cost effective Point-of-Care rapid tests.
LEARN MORE
-
-
Global Leader
Commitment to Quality & Care
Our mission is to build a healthier tomorrow for Community.
LEARN MORE -
Distribution
We Distribute Worldwide
Indonesia, France, Albania, Austria,Ukraine, Chad, Thailand, Nigeria...
LEARN MORE -




Our Products
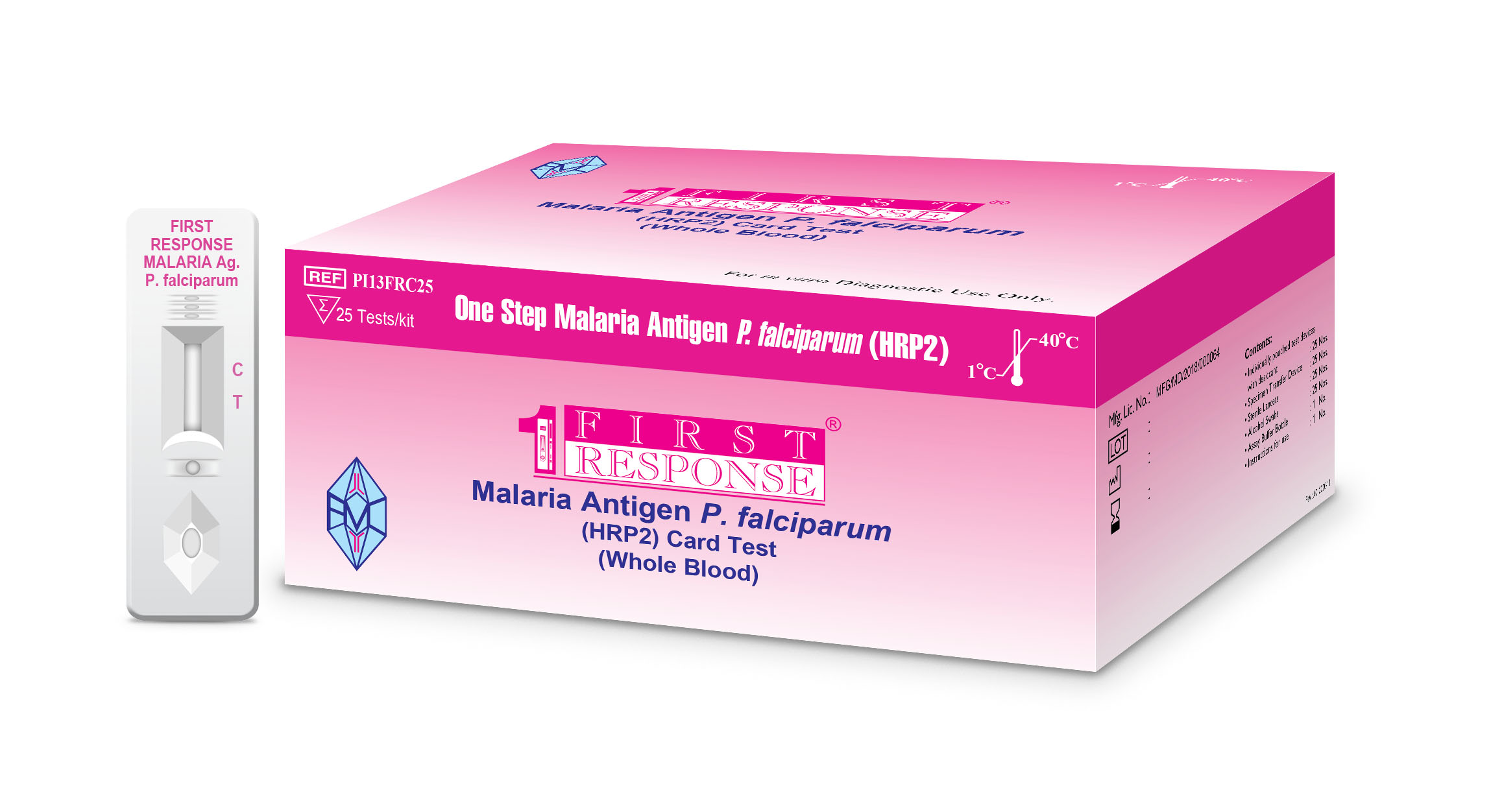
First Response® Malaria Antigen P.falciparum (HRP2) Card Test
First Response® Malaria Antigen P.falciparum (HRP2) Card Test is intended to be performed by trained users (In either laboratory or point of care settings) as qualitative screening in vitro diagnostic test for detection of P. falciparum specific HRP2 antigen.
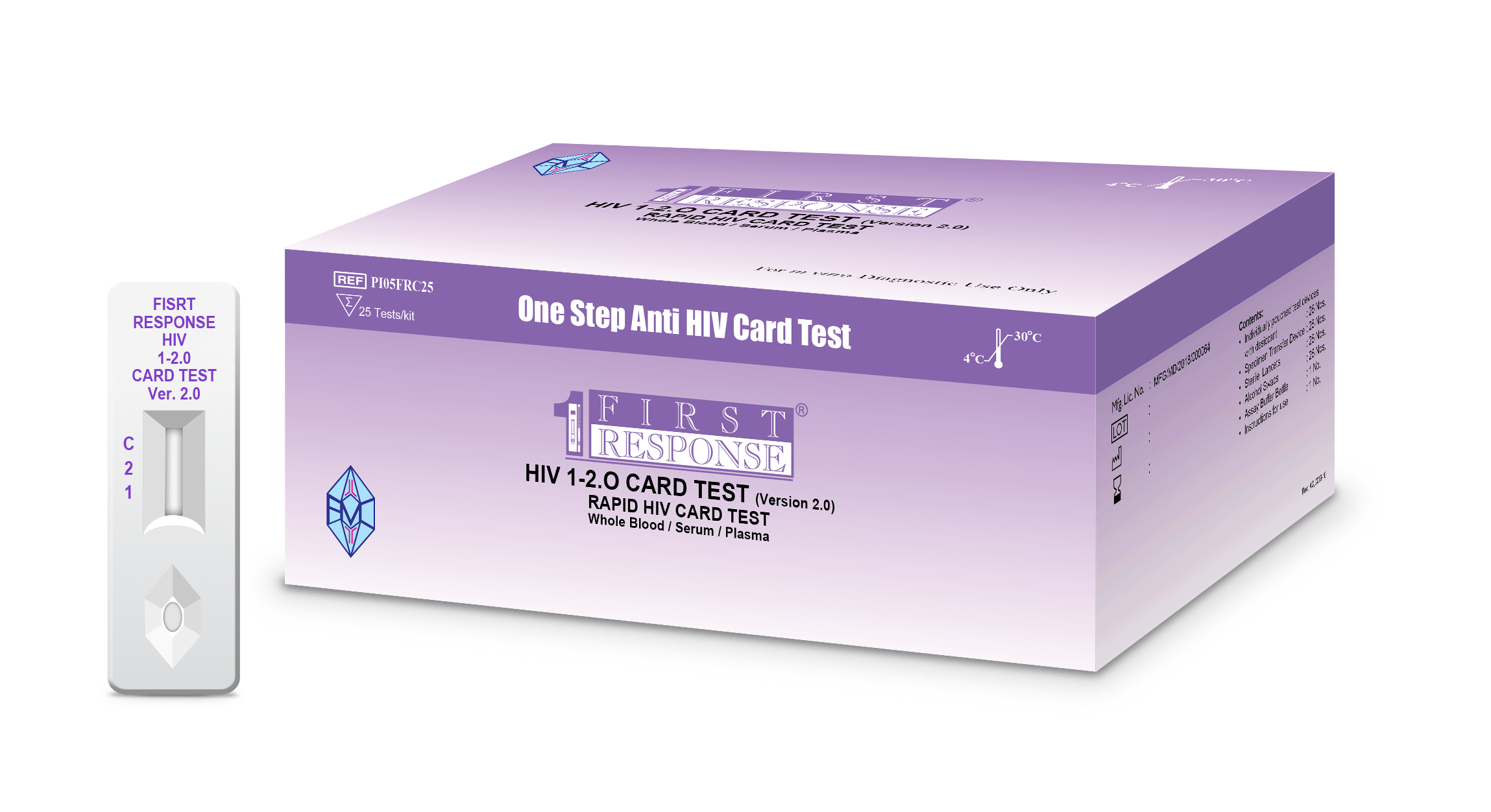
First Response® HIV 1-2.O Card Test (Version 2.0)
First Response® HIV 1-2.O Card Test (Ver. 2.0) is intended for use by healthcare professionals and qualified laboratory personnel. It is a rapid, qualitative screening, in vitro diagnostic test for detection of antibodies specific to HIV-1 (including Group O) and HIV-2 in human serum, plasma or venous and capillary whole blood.
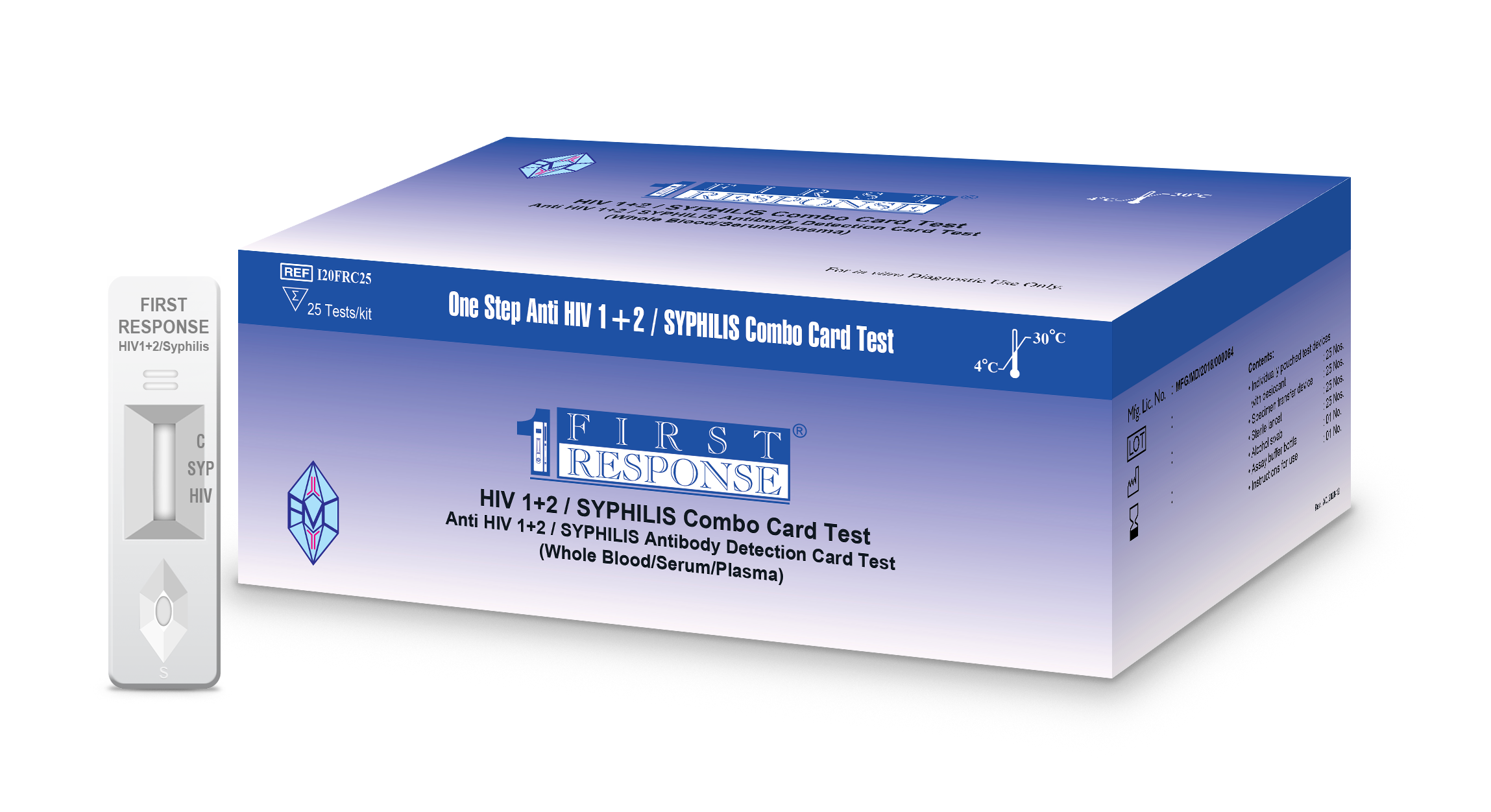
First Response® HIV 1+2/Syphilis Combo Card Test
First Response® HIV 1+2 / Syphilis Combo Card Test is intended for use by healthcare professionals and trained user. It is a rapid, qualitative screening, in vitro diagnostic test for the detection of antibodies (IgG & IgM) specific to HIV (type 1 & 2) and Treponema pallidum in human serum, plasma or venous and capillary whole blood.

First Response® Syphilis Anti TP Card Test
First Response® Syphilis Anti-TP Card Test is intended for use by healthcare professionals and qualified laboratory personnel. It is a rapid, qualitative, screening, in vitro diagnostic test for the detection of antibodies of all classes specific to Treponema pallidum in human serum, plasma or Venous or capillary whole blood.
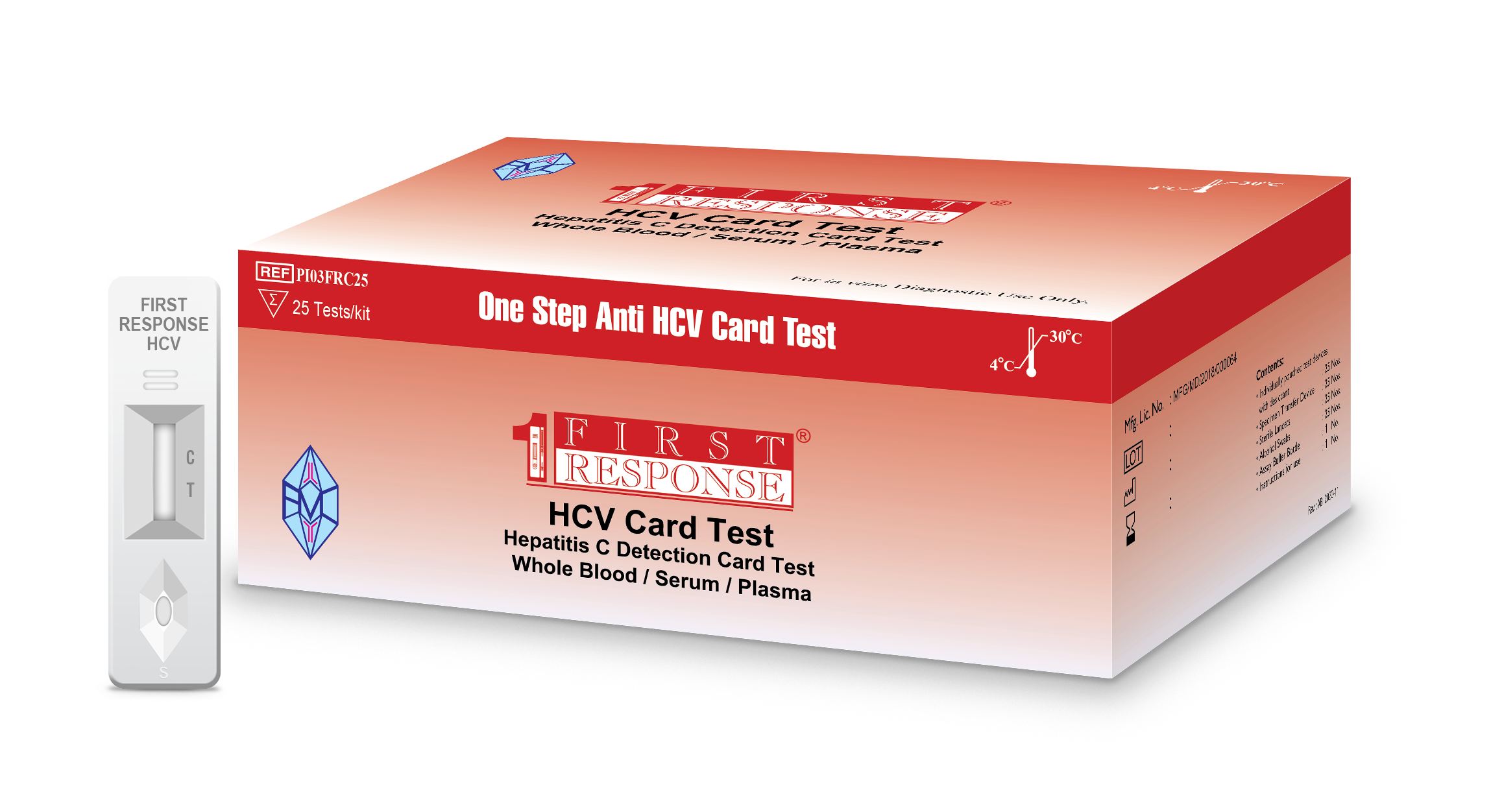
First Response® HCV Card Test
First Response® HCV Card Test is a chromatographic immunoassay for the qualitative detection of the antibodies against hepatitis C virus (HCV Ab) in human serum, plasma or whole blood (venous & capillary blood) specimens.




















